2023
- May 21st. Evolution of alternative genetic codes in endosymbionts.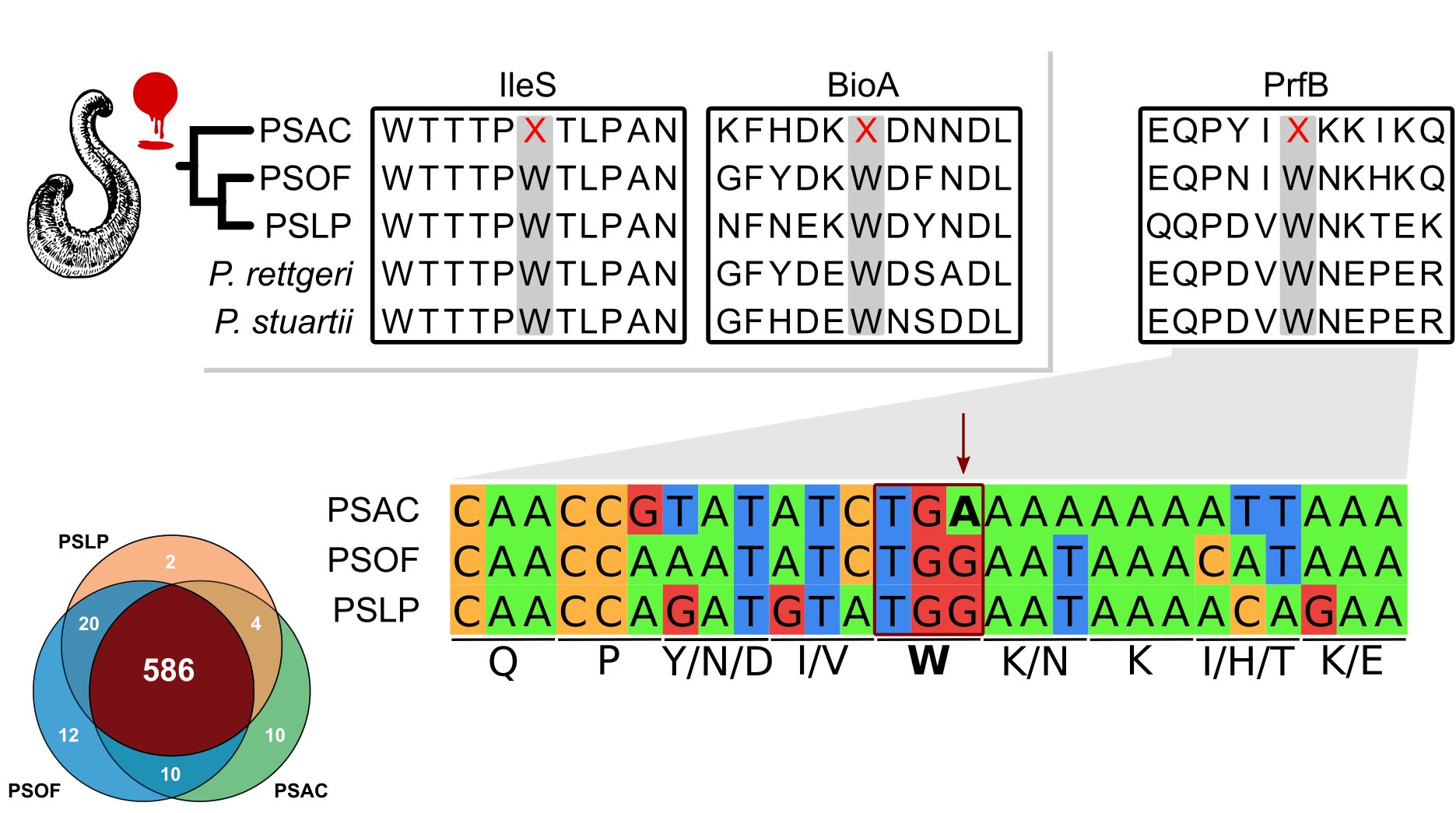
Obligate vertically-transmitted endosymbionts are weird... Given their particular lifestyle and mode of inheritance, they have evolved all sorts of particular genomic features (small to tiny genomes, biased nucleotide composition, among others). These sort of weird critters not only live inside insects, but can be found widely spread across invertebrates with a nutrient restricted diet. Strict blood-feeding leeches have evolved these sort of symbionts, but little is known about their nature, identity and genomics. Thus, we set to further explore the Providencia-Haementeria symbiosis by expanding the genomic knowledge of this symbiont lineage.
We found that similarly to other long-term vertically transmitted symbionts, the Providencia siddallii ancestor evolved a small gene-dense mobile element-free genome (so far, pretty standard, right???). Most surprisingly, we found that a "rare" genetic code reassignment (11->4) evolved in the symbiont lineage associated to the mexican leech H. acuecueyetzin (fun fact! the specific epithet means "leech" in Nahuatl). This reassignment means that the STOP codon 'UGA' is now coding for tryptophan! Moreover, features form the symbiont genomes suggest an early stage of reassignment and an ancestral propensity for the P. siddallii bacteria to undergo such a reassignment, e.g. the ancestral accumulation of 'UGA' codons in essential genes. This work was a great collaboration with my friends and long-lived colleagues Dr. Alejandro Oceguera Figueroa (Biology Institute, UNAM, Mexico) and Dr. Sebastian Kvist (Natural History Museum, Stockholm, Sweeden).
2022
- September 4th. Aphids, aphids, and more aphids!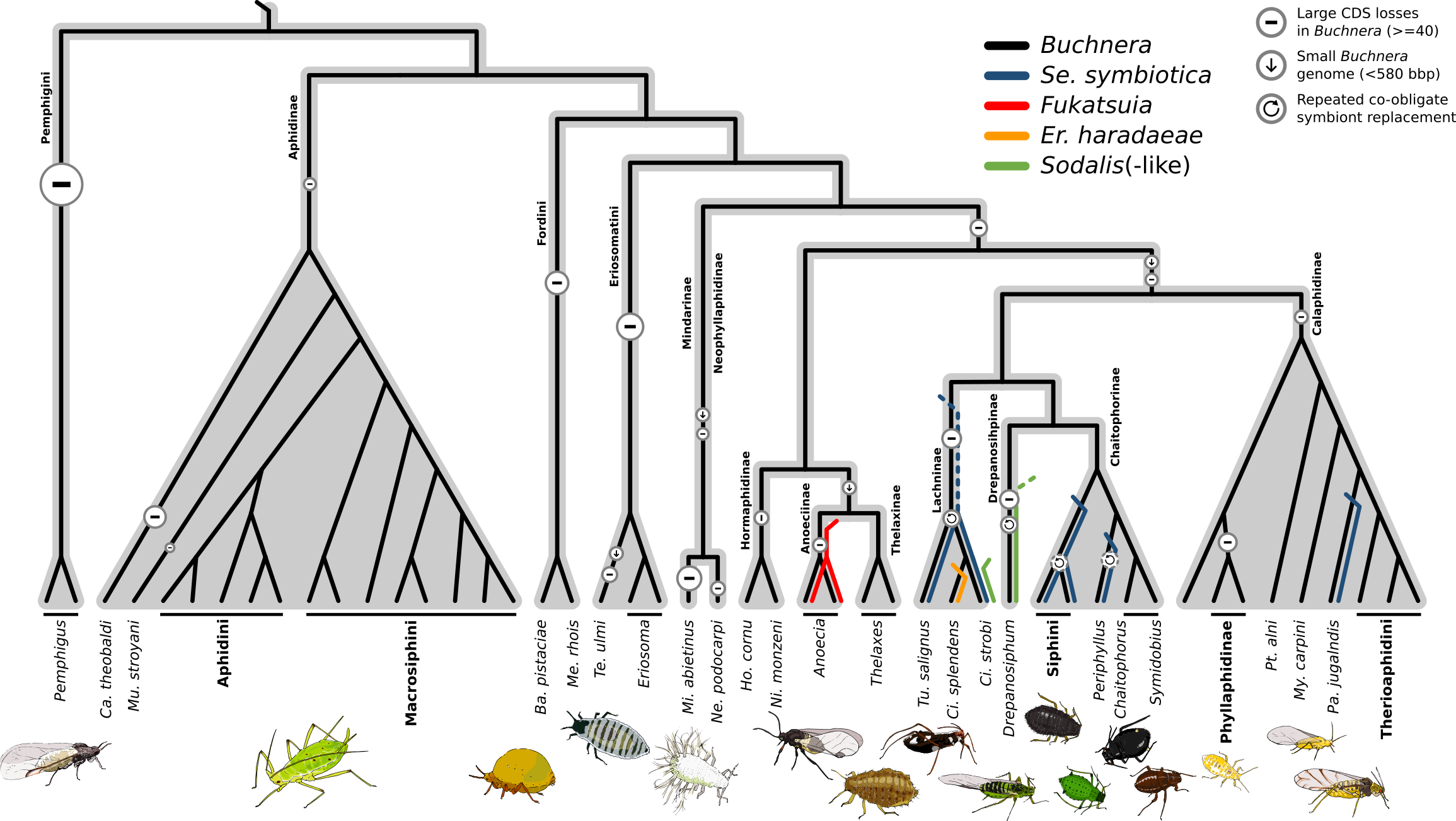
After many years of work, our paper on co-obligate symbioses across aphids is finally out in bioRχiv!. Aphids are a large family of hemipterans with around 5,200 species organised into 23 extant subfamilies. Most work relating to endosymbionts has been done on the Aphidinae (60% of diversity), where aphids typically host a single nutritional symbiont, Buchnera. However, previous evidence shows that, similarly to adelgids and pseudococcids, some aphids can host more than one obligate nutritional symbiont. These co-obligate symbioses exist in Lachninae and Periphyllus spp., but we know little of these associations across aphids.
In this work, we sought to explore the nature and evolution of these associations. To this goal, we performed 16S amplicon sequencing on a total of 223 aphid samples (147 species, 12 subfamilies). In addition, we newly sequenced the symbionts of 25 aphid species & re-assembled 20 more. We observed that, despite large-scale gene loss in Buchnera often being associated to the acquisition of a new co-obligate symbiont, these two processes can be decoupled: in fact, only a few losses can trigger the establishment of new multi-partite nutritional symbioses. In addition, B vitamins (mainly riboflavin, seem at the core of the evolution of these new co-obligate symbioses (in aphids), with the biosynthesis of biotin often also being taken-over by the new symbiont. In fact, some even bring a new nutrient to the mix, thiamin.
New co-obligate symbionts have most commonly evolved from bacterial taxa known to be facultative endosymbionts of aphids, stressing the facultative-to-obligate lifestyle shift. In conclusion, co-obligate symbioses have evolved anew at least 6 times across aphids, thus existing in upwards of 11% of aphid species (the number is way higher is you do not count the Aphidinae subfamily □).
- June 25th. On your face! New work published on face mites!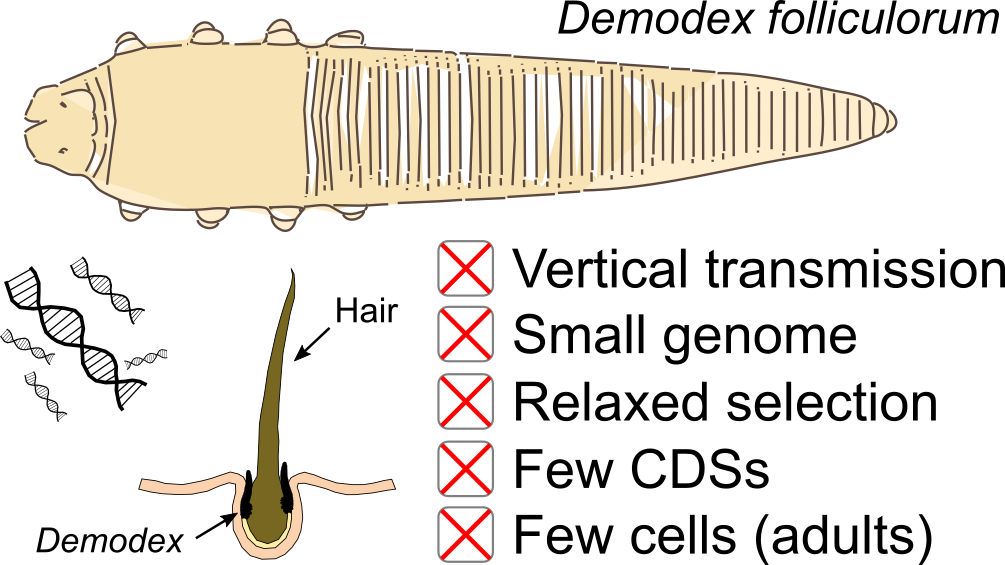
Demodex folliculorum mites, members of the arachnid class, inhabit the skin of (almost) all humans and other mammals. They spend their whole lives as living as symbionts, cannot survive outside their host, and rarely have the opp. to outbreed. Sounds familiar? You're right! just like bacterial endosymbionts do!
We found the genome of Demodex folliculorum holds several features of historical and ongoing genome reduction: 1) small compact genome, 2) widespread relaxed selection on genes, and 3) a staggering low amount of CDSs. In addition, rearrangement and changes in their Hox genes likely impacts their unique body plan such as the anterior and dorsal position of the penis and lack of segmentation. Lastly, the work shows Demodex is generally vertically transmitted and it indeed has an anus! and therefore have been wrongly blamed for many skin diseases attributed to the accumulation of faeces during their lifetime before release when Demodex died. This collaborative work has been published in Molecular Biology and Evolution
- June 13th. New work published!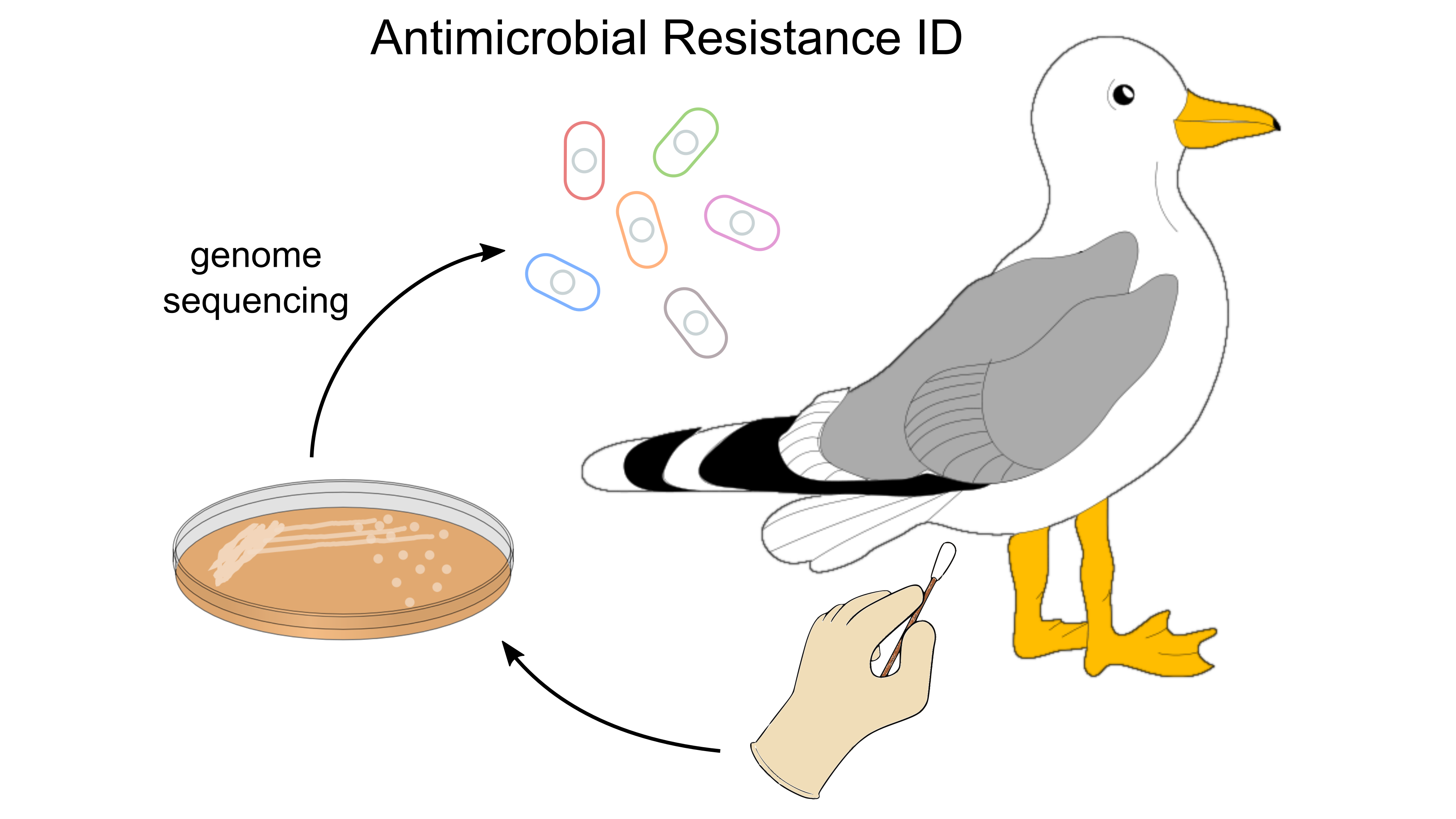
If you are interested in cloacal swabs, bacterial culturing, and/or antimicrobial resistance, we've got you covered! In this collaborative venture published in Ecology and Evolution, isolated bacteria from Yellow-legged gull chicks were sequenced and screened for antimicrobial resistance genes. We found antimicrobial resistance genes in many Escherichia, Enterobacter, Citrobacter, Hafnia, Klebsiella, and Proteus. Importantly, this study revealed an increase in the diversity of resistant enterobacteria over time (hatching to fledging) in chicks. This increase in diversity was accompanied by an increase in the proportion of individuals carrying bacteria with antimicrobial resistance genes. There are three possible explanations for this observation: 1) food supply change, 2) parents, through time, might forage in different areas, and/or 3) an increasing number of exchanges with other chicks.
2021
- December 1st. A long pandemic year, but fruitful nonethelessDefinitely a rough year: further lockdowns, difficulties doing field work, and a lab move. Nonetheless, some good things actually managed to happen during this time.

Back in September, Kevin (a former co-directed master student from the Royal Ontario Museum) joined me at DoME. Kevin will be working with me to explore the ins and outs of bacterial symbioses across strict blood-feeding leeches. In addition, Luka Močivnik from the University of Ljubljana joined me at DoME for one month to work on a pipeline for the phylogenomics of the Niphargus, the largest genus of freshwater amphipods (Stay tuned!). Lastly, and despite complications and some deaths, all the leech populations have been successfully moved to the new building.
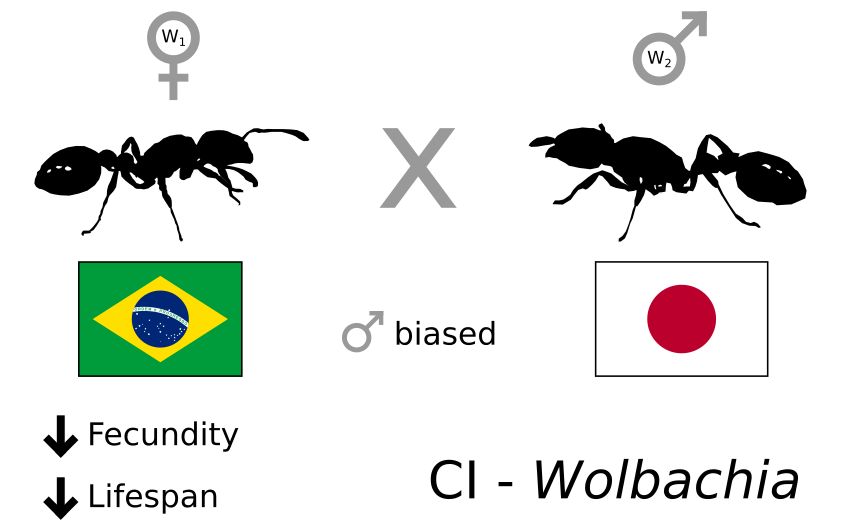
On the publication side of things, a total of two works were completed in this year. The first has been published in Evolution, and was the result of a recurring collaboration with Jan Oettler and Eva Schultner. In this work we explored the Wolbachia-induced reproductive manipulation between two populations of the ant species Cardiocondyla obscurior infected by two distinct Wolbachia strains. Crosses of infected queens of a Brazilian population with infected male individuals from a Japanese population, resulted in male-biased offspring and reduced lifespan and fecundity of Brazilian queens. This work shows the role of Wolbachia-induced cytoplasmatic incompatibility in creating reproductive isolation between populations of a widely-distributed ant species.
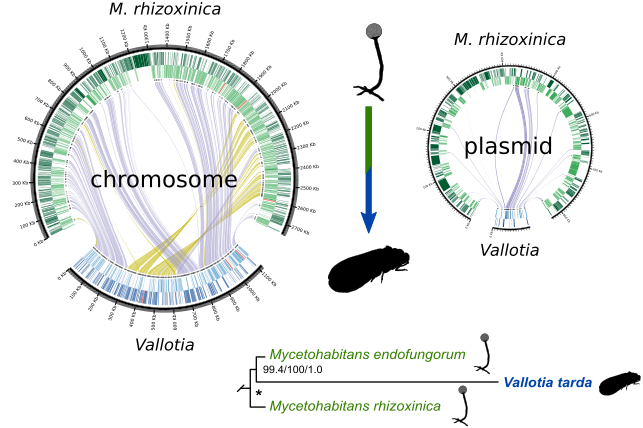
The second work has been published in The ISME Journal, and resulted from a collaboration with current and former members of my new workplace. We performed whole-genome sequencing to analyse the co-obligate symbiotic system of the adelgid Adelges laricis/tardus. We found that both endosymbionts, Profftia and Vallotia (now Mycetohabitans vallotii), show characteristics of evolutionary "young" endosymbionts. On the other hand, they show strong metabolic complementarity in production of essential amino acids and B vitamins. Despite the genome reduction observed in M. vallotii, synteny is well conserved when compared to other Mycetohabitans, with the chromosome + plasmid organisation of its genome maintained. Most notably, we show that M. vallotii likely derived from a fungus-associated endosymbiont, a hitherto unusual evolutionary trajectory for a nutritional insect-associated endosymbiont.
2020
- June 20th. Coronavirus, lockdowns, and getting slowly back to the lab (or trying to..).
Well, what to say about the first couple of months of 2020. I finally relocated to Vienna, after finally getting a Marie Skłowdoska-Curie IF fellowship (LEECHSYMBIO). With this grant, I finally had the opportunity to start my very own independent line of work exploring the highly overlooked nutrition-based symbioses between leeches and their bacteriocyte-associated bacteria. After officially starting with my new position at the CMESS in mid-January, I started planning the experiments, I received my leech samples, got my work computer, and went through all the lab courses to prepare for my project. Next, mid-March, the COVID-19 lockdown officially started in Vienna. So, I had to stop all work and take my work computer home. Ever since then, it has been full-time home office working on past projects.

As for hundreds of other Early Stage Researchers (ESRs), my project is now threatened by the now 3 months I have been unable to work on my project. While other funders such as EMBO, the German DFG, and the Portuguese FCT announced costed extensions early on (i.e. project extensions with accompanying salary for the ESR), the Marie Skłowdoska-Curie Actions (MSCA) did not. After months of a petition I launched (which has now raised over 1,500 signatures), an open statement published by the Marie Curie Alumni Association (MCAA), a letter to President von der Leyen co-signed by 33 Marie Skłowdoska-Curie fellows, and a survey launched with through the MCAA and passed on to the MSCA, we continue to be denied costed extensions. While the European Commission (EC), President von der Leyen, Commissioner Mariya Gabriel, the REA, and the MSCA have all expressed open support for science, tangible support in the form of costed extensions for ESRs has been lacking: we are being hung out to dry. With more and more national and local funders announcing costed extensions, the European Commission has made it very clear we are dispensable to them and they are willing to risk our careers, projects, mental health and personal lives. I have never been more disappointed and ashamed to be part of such an institution whose priorities have become clear with this pandemic. You can read more in the article I wrote for EuroScientist, coverage done by reporter Pola Lem here and here, and a brief coverage of the lack of costed extensions by reporter Julie Gould for Nature's coronapod (min 22:30).
2019
- December 7th. Another November passed, and two works published!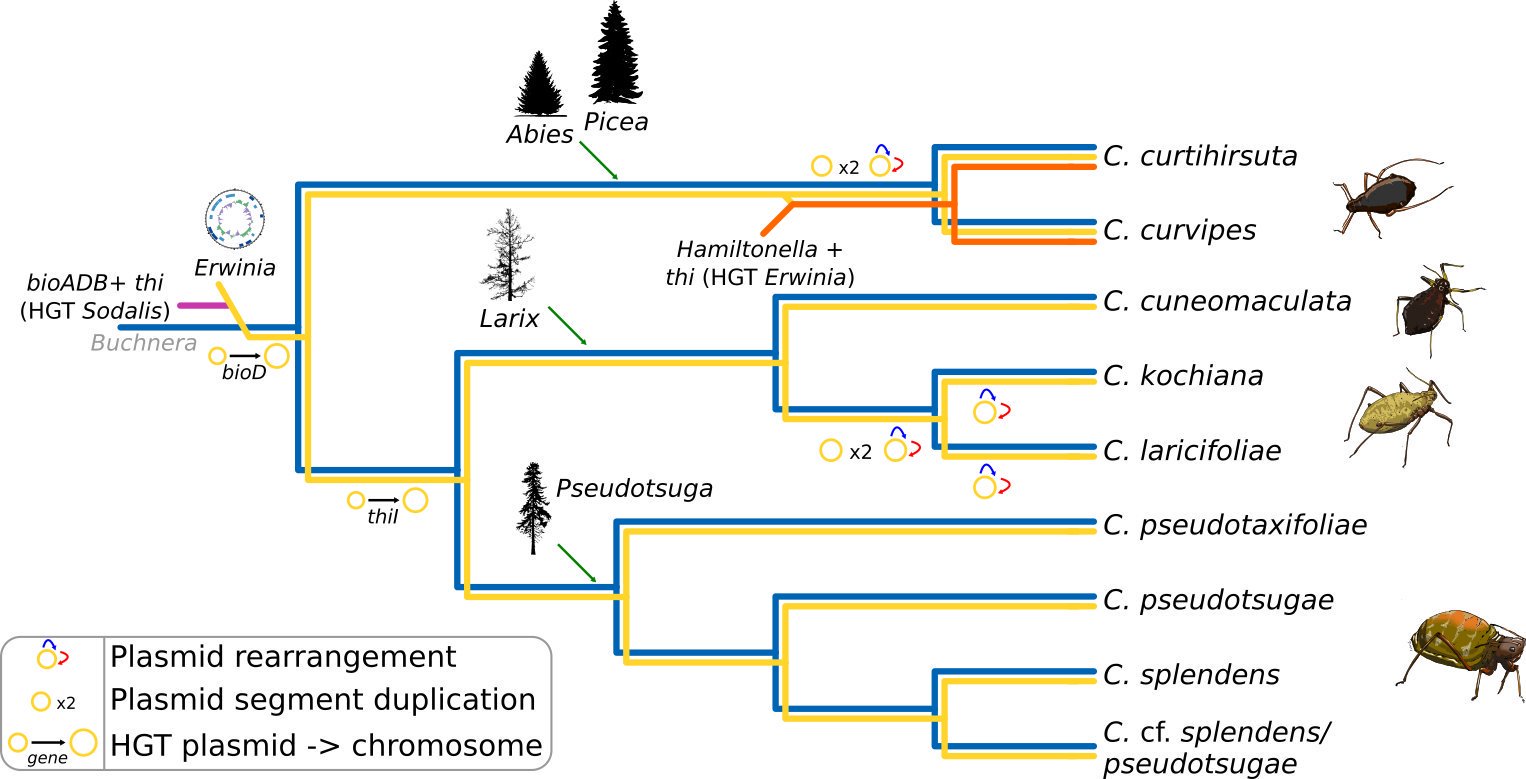
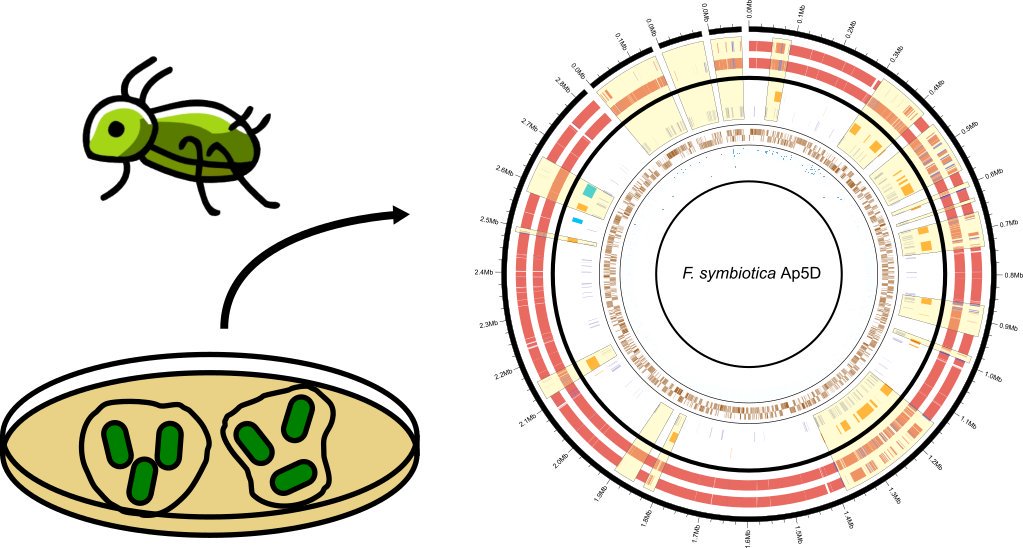
This work was the result of a many many months of exhausting genome assembly, annotation, comparative genomics, and phylogenetics. In this work, we analysed the genome of a newly described co-obligate endosymbiont, Ca. Erwinia haradaeae, of a particular group of aphids within the Cinara genus (Manzano-Marín et. al. 2020). We found that Ca. Erwinia haradaeae, similarly to Buchnera, underwent an early genomic reduction and settled as a co-obligate endosymbiont early before the diversification of the group, likely replacing a pre-existing co-obligate Serratia symbiotica. As expected, they complement Buchnera in key nutritional pathways (riboflavin and biotin) to supply the aphid host with these vitamins. Most significantly, we found these most relevant genes have been horizontally acquired form a Sodalis-like bacterium, highlighting the important role horizontal transfer of genes can play in the establishment and maintenance of beneficial host-microbe associations.
This second work, was the product of a really cool collaboration (I played a very small part) with the Oliver lab and co. (Patel et. al. 2019). Briefly, a facultative Ca. Fukatsuia symbiotica from the pea aphid was cultured in lepidopteran cells and isolated for whole genome sequencing (cool). Full genome analyses and comparative genomics against a co-obligate strain of the same species from a Cinara aphid was performed, and they were not that different. Finally, while Ca. Fukatsuia symbiotica and Ca. Hamiltonella defensa have a strong tendency to co-occur, metabolic complementarity (inferred from available genomes) does not seem to be the basis for this observed characteristic.
- June 27th. Back from the GRS/GRC Animal-Microbe Symbioses and elected as next co-chair for the 2021 GRS!

Back home from the GRS/GRC Animal-microbe symbioses meeting of 2019, great as always. It certainly felt long, but the science and networking was great. I ran into some friends which I barely get to see and had a lot of interesting discussions during the poster sessions and social hours. This year there were a ton of symbiotic systems being presented as both posters and talks: the Hawaiian bobtail squid and their light organ endosymbionts, phloem feeder insects and their nutritional obligate bacteria, the fruit fly and Wolbachia, the defensive symbionts of digger wasps (A.K.A. beewolves), among many others. I presented my work on the serial horizontal gene transfer and the origin/establishment of new obligate nutritional endosymbionts in Cinara aphids (doi: 10.1101/556274). Additionally, I am very happy that I, along with Nancy Obeng, was elected to be co-chair for the upcoming 2021 GRS Aminal-microbe symbioses! (which will hopefully happen in Italy, fingers crossed).
